Home / 응용분야 / 응용분야 전체
-

Cultured Meat: Sustainable Innovation & Role of High-throughput Automated Cell Counter
With advancements in biotechnology and a growing concern for the environmental impact of traditional animal farming, cultured meat has emerged as a promising solution that could revolutionize the way we produce and consume protein. Cultured meat is produced by culturing animal cells in a controlled environment, without the need for raising and slaughtering animals. NanoEntek’s high-throughput automated cell counters, EVE™ HT and EVE™ HT FL, can accelerate the production of cultured meat. NanoEntek’s high-throughput automated cell counters, EVE™ HT and EVE™ HT FL, can accelerate the production of cultured meat 1. What is Cultured Meat? Cultured meat, also known as lab-grown or cultivated meat, is produced by culturing animal cells to create meat products without the need for traditional livestock farming and slaughtering. This innovative approach involves isolating stem cells from animals such as cows or chickens and providing them with the necessary nutrients and environmental conditions to proliferate and differentiate into muscle, fat, and connective tissue cells. The resulting product closely resembles conventionally produced meat but offers significant advantages in terms of environmental sustainability, animal welfare, and potential health benefits. 2. Emerging Necessity of Cultured Meat (1) Continuous Growth of World’s population As of January 25, 2024, the global population has surpassed 8 billion and is projected to reach nearly 9 billion by 2050, according to real-time statistics from Worldometers. This population growth, coupled with shifting dietary habits towards increased meat consumption, especially in emerging economies, exacerbates ethical and environmental concerns associated with traditional meat production. (2) Growing demand for Meat Global meat production has surged from 70.57 million tons in 1961 to 337.18 million tons in 2020, driven by population growth and economic development. The Food and Agriculture Organization of the United Nations predicts that meat demand will double by 2050. Livestock farming is a major contributor to greenhouse gas emissions, accounting for 14.5% of global emissions, with cattle farming being the largest emitter due to methane release. 3. Benefit of Cultured Meat Cultured meat offers a sustainable solution to the environmental and ethical challenges posed by conventional meat production. With up to 92% lower greenhouse gas emissions, 90% less land use, 98% less soil acidification, and 94% less air pollution compared to conventional beef, cultured meat addresses key sustainability issues. Additionally, its production is not limited by geographical constraints, enhancing food security and production efficiency while reducing the risk of zoonotic diseases and antimicrobial resistance.4. How are cultured meat made? Cultivated meat production embodies the principles of biomimicry, a discipline that draws inspiration from natural processes to develop innovative technologies. In the context of cellular agriculture this entails replicating the inherent cellular growth mechanisms found in animals outside of their living organisms. The manufacturing process starts by isolating and banking stem cells from an animal, which can self-renew and differentiate. These cells are then cultivated in bioreactors, often referred to as cultivators, in large quantities. These cells are nourished with a nutrient-rich medium containing amino acids, glucose, vitamins, salts, and growth factors to stimulating the conditions inside the animal’s body.By adjusting the composition of the medium and providing cues from a scaffolding structure, the immature cells are prompted to develop into various tissues like skeletal muscle, fat, and connective tissues, which are the components of meat. Once the cells have differentiated, they are harvested, processed, and packaged into final products. This entire process typically takes between 2 to 8 weeks, depending on the type of meat being cultivated. 5. The Role of High-throughput Automated Cell Counter(1) NanoEntek’s EVE™ HT Series: Transforming Bioprocess Efficiency with Superior Throughput, Minimal Sample Requirements, and Swift Measurement Times The starting cell type ultimately influences many of the downstream variables of the bioprocess, including timelines and differentiation strategies. Accurate measurement of cell density during the initial seeding process is pivotal. A slight error in the seeding density can profoundly affect subsequent processes. Additionally, in bioreactors, it is necessary to culture a large number of cells rapidly. Therefore, periodic sampling and analyzing a substantial quantity of samples is essential in bioprocessing.From this perspective, NanoEntek’s high-throughput automated cell counters, EVE™ HT and EVE™ HT FL, have an edge over other conventional high-throughput cell counters in terms of throughput, required sample volume, and measurement speed.♦ Throughput Conventional high-throughput automated cell counters have the capacity of measuring only up to 24 samples maximum. However, EVE™ HT and EVE™ HT FL is capable of measuring up to 48 samples per each test, enhancing the productivity of the R&D and manufacturing process. Researchers and operator could as a result, easily and quickly find the optimal condition in extensive experiment with different combination of parameters. NanoEntek's EVE™ HT FL Multichannel counting plate: Up to 48 fluorescence-stained samples to be loaded at once.♦ Required Sample Volume Whereas conventional high-throughput cell counters demand up to 500 µL of sample per test, EVE™ HT and EVE™ HT FL stand out by requiring only a minimal 20µL per test. This significant reduction not only showcases EVE™ HT and EVE™ HT FL’s exceptional efficiency but also underscores their ability to conserve valuable samples ♦ Measurement Time For a batch of 48 samples, the EVE™ HT series completes the measurement process in only 3 minutes. In comparison, other equipment can take up to around 210 minutes. This represents a staggering time efficiency gap of nearly 70-fold, firmly establishing the EVE series as a leader in time efficiency.EVE™ HT FL is much more faster and requires substantially low sample volume compared to competitors.Given these compelling advantages, NanoEntek’s EVE™ HT and EVE™ HT FL high-throughput automated cell counters offer high efficiency and accuracy for the initial bioprocess stages.(2) The Integral Role of EVE™ HT and EVE™ HT FL in Cultured Meat ManufacturingNanoEntek’s high-throughput automated cell counters, the EVE™ HT and EVE™ HT FL, are indispensable tools in the cultured meat production process, especially during the initial manufacturing stages. These devices are pivotal in monitoring and analyzing cell cultures, ensuring their health and viability throughout the production cycle.By automating cell counting, these counters streamline operations, saving time and reducing labor while minimizing the risk of human error. Furthermore, they furnish researchers with precise and dependable data on cell growth and proliferation.High-throughput automated cell counters are essential for assessing the quality and consistency of cultured meat products, ensuring that each batch meets the desired specifications. Additionally, these counters play an essential role in process optimization, enabling researchers to detect and rectify potential issues at the onset of the production cycle.EVE™ HT and EVE™ HT FL measure up to 48 samples per test using only 20µL of sample, completing a batch measurement of 48 samples in just 3 minutes.NanoEntek's high-throughput automated cell counters, EVE™ HT and EVE™ HT FLConclusionIn conclusion, cultured meat has the potential to revolutionize the food industry by offering a sustainable, ethical, and healthy alternative to traditional meat. NanoEntek's high-throughput automated cell counters, EVE™ HT and EVE™ HT FL, are instrumental in realizing this potential by providing researchers with the tools they need to produce cultured meat products that meet the highest standards of quality and consistency.Overall, the development of cultured meat represents a significant step forward in addressing some of the most pressing challenges facing the food industry today. By harnessing the power of technology and innovation, we have the opportunity to create a more sustainable, ethical, and healthy food system for future generations.

-
EVE™ HT: Efficient, Accurate, and High-Throughput Solutions for Stem Cell analysis
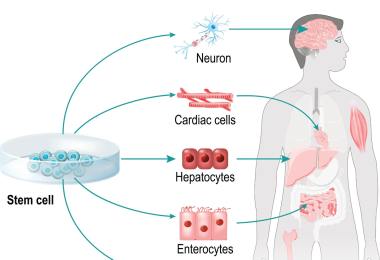
Summary Stem cell therapy involves using viable human stem cells to regenerate tissues and treat various diseases. It encompasses embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and adult stem cells. EVE™ HT uses trypan blue for quick and accurate viability evaluation in up to 48 samples within 3 minutes.Cell Therapy and its classificationCell therapy uses living cells to regenerate and restore damaged or diseased cells. It has applications in drug development, toxicology testing, and biomarker research. The three main types are tissue cell therapy, immune cell therapy, and stem cell therapy. The following content will primarily focus on stem cell therapy. The new treatment uses stem cell from various sources to regenerate cell or tissue types. What is Stem Cell therapy? Stem cell-based therapies are defined as any treatment for a disease or a medical condition that fundamentally involves the use of any type of viable human stem cells including embryonic stem cells (ESCs), iPSCs and adult stem cells for autologous and allogeneic therapies. Stem cells offer the perfect solution when there is a need for tissue and organ transplantation through their ability to differentiate into the specific cell types that are required for repair of diseased tissues. [1] Where Can I use Stem Cell therapy? Stem cells serve as the source for all cells, possessing undifferentiated characteristics, self-replication abilities, and the potential for differentiation. This differentiation function allows the treatment of damaged organs or tissues by promoting division and indefinite duplication. Stem cells are categorized into adult stem cells (AS cells), induced pluripotent stem cells (iPS cells), and embryonic stem cells (ES cells).Stem cell therapy, harnessing the capacity to differentiate and multiply into diverse cells, has been extensively studied and developed for various diseases, facilitated by technological advancements. Many developed countries offer policy support, recognizing it as a key component of the medical industry. Stem cells also serve as biomarkers to monitor post-treatment effectiveness and as cell treatments for incurable diseases.Through differentiation and proliferation, stem cells can be injected into patients to replace damaged cells, fostering tissue development in cell therapy. Additionally, stem cells play a role in enhancing the efficiency of new drug development by identifying candidates and assessing their efficacy. Stem cell therapy has shown effectiveness in treating a myriad of conditions, including burns, cardiovascular diseases, muscle diseases, vision impairment, and Alzheimer's disease. Additionally, stem cells hold promise for addressing challenging diseases such as spinal cord injuries, dementia, Crohn's disease, Parkinson's disease, Lou Gehrig's disease, and multiple sclerosis.Among various stem cell types, hematopoietic stem cells (HSCs) play a crucial role in producing red blood cells, white blood cells, platelets, and immune cells like T-lymphocytes and B-lymphocytes. Hematopoietic stem cell transplantation (HSCT) is a rapidly growing procedure worldwide, finding application in treating conditions such as aplastic anemia, acute leukemia, and solid cancers. EVE™ HT: Efficient, Accurate, and High-Throughput Solutions forWhen working with tissue-derived cells, particularly stem cells, accurate assessment of cell concentration and viability is essential for maintaining quality throughout subsequent procedures. A High Throughput Trypan Blue-Based Cell Counter, such as the EVE™ HT, is a valuable tool in the manufacturing process of stem cell therapy products, offering specific capabilities that contribute to the overall efficiency and quality control.EVE™ HT EVE™ HT is an automated cell counter that uses trypan blue for high-throughput measurements. In less than 3 minutes, it can accurately measure up to 48 samples, making it an efficient solution for busy laboratories or industries. The efficiency is maximized through the use of a disposable 48 channel plate.Here's a detailed explanation of how it can be utilized:Why is evaluating viability of cells important in Stem cell therapy?Therapeutic Efficacy: Ensuring PotencyThe therapeutic efficacy of stem cell therapies is directly linked to the potency of the administered cells. Only viable and functional stem cells have the potential to contribute to tissue regeneration or repair. Monitoring cell viability guarantees that the therapeutic product contains a sufficient number of viable cells capable of carrying out the intended therapeutic functions.Patient Safety: Minimizing RisksAdministering non-viable or compromised stem cells to patients can pose serious risks. Non-viable cells may not survive after transplantation, leading to treatment failure and potential harm to the patient. By assessing viability during the manufacturing process, the risk of administering ineffective or harmful cell products can be minimized.Consistency and Reproducibility: Quality ControlViability measurements contribute to maintaining consistent product quality. Quality control measures ensure that each batch of stem cell therapy meets predefined standards. Consistency in viability levels across batches enhances reproducibility, allowing for reliable therapeutic outcomes and facilitating regulatory compliance.Optimizing Dosing: Accurate Cell DosingDetermining the appropriate cell dose for each patient is crucial for the success of stem cell therapy. Viability data is essential for calculating the actual number of viable cells in a given dose, allowing clinicians to administer an optimal and standardized cell dose tailored to individual patient needs.Cost-Effectiveness: Efficient Resource UtilizationMonitoring cell viability helps avoid wasting resources on non-viable cells. Inefficient use of resources, such as culture media, supplements, and laboratory personnel time, can be minimized by eliminating non-viable cell populations early in the manufacturing process. This contributes to cost-effectiveness in the production of stem cell therapies.Post-Transplantation Outcomes: Predicting In Vivo PerformanceThe viability of stem cells before transplantation can influence their survival and behavior in the patient's body. Monitoring viability helps predict post-transplantation outcomes, providing valuable information for clinicians and researchers to optimize treatment protocols and improve long-term therapeutic benefits.Reference[1] Current state of stem cell-based therapies: an overview, Stem Cell Investigations

-
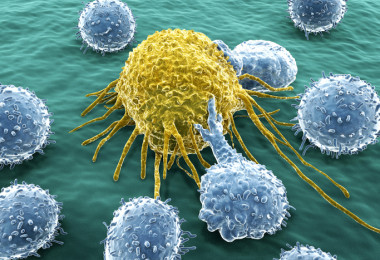
EVE™ HT FL: Elevating CAR-T Therapy through High Throughput Precision
Immune cell therapy, particularly CAR-T cell therapy, utilizes the body's immune system to combat diseases, primarily cancer. CAR-T cells are genetically modified T cells designed to target and destroy cancer cells. In the manufacturing process of CAR-T, high-throughput fluorescence Cell Counters like EVE™ HT FL play a crucial role. They assess cell viability, enumerate CAR-T cells, ensure quality control of starting material, optimize culture conditions, determine optimal harvesting time, detect contaminants, and facilitate data documentation for regulatory compliance, enhancing efficiency and quality in CAR-T manufacturing. In summary, EVE™ HT FL enhances efficiency and quality in CAR-T manufacturing, contributing to the success of this personalized immunotherapy. Cell Therapies: Tissue cell therapy, Immune cell therapy, and Stem cell therapy Cell therapies encompass tissue cell therapy, immune cell therapy, and stem cell therapy. In biomedicine, they stand as distinct fields, with cell therapy further divided into tissue-cell therapy, immune cell therapy, and stem-cell therapy. Gene therapy, on the other hand, is categorized into immune cell gene therapy and stem cell gene therapy. This discussion focuses on immune cell therapy, specifically CAR-T, and how high-throughput fluorescence Cell Counters such as EVE™ HT FL are applied in CAR-T manufacturing. Biomedicine Classification What is immune cell therapy?Immune cell therapy, also known as immunotherapy, is a medical approach that utilizes the body's immune system to combat diseases, particularly cancers and immune-related disorders. This personalized treatment involves extracting a patient's immune cells, genetically modifying them in the laboratory to enhance their targeting abilities, and reintroducing them into the patient's bloodstream. Its applications extend to new drug development, toxicology testing, and biomarker research. Notable examples include CAR-T cell therapy, where engineered T cells specifically target and destroy cancer cells. Immune cell therapy offers precision medicine tailored to an individual's immune profile and has shown remarkable success in certain cancers, opening new possibilities for treating previously challenging conditions. Ongoing research aims to expand its applicability and optimize safety and efficacy. What is CAR-T cell? CAR-T cells are T cells that undergo genetic modification to create an artificial T-cell receptor. This innovative immunotherapy aims to enhance the ability of T cells to identify and eliminate cancer cells. The process involves extracting T cells from individuals, altering them genetically, and then reintroducing the engineered CAR-T cells into patients to target and destroy tumors more effectively. These CAR-T cells can either come from the patient's own blood (autologous) or from a healthy donor's T cells (allogenic). To ensure safety, CAR-T cells are designed to specifically recognize antigens present on tumors but not on healthy cells. EVE™ HT FL: Efficient, Accurate, and High-Throughput Solutions for CAR-T productsWhen working with cells like PBMCs or T-cells, accurate assessment of cell concentration and viability is essential for maintaining quality throughout subsequent procedures. Cell counting in these kinds of samples becomes a challenging task due to common contaminants like red blood cells and non-cellular debris, introducing complexities such as time consumption, subjectivity, and a higher likelihood of errors.EVE™ HT FL When working with cells like PBMCs or T-cells, accurate assessment of cell concentration and viability is essential for maintaining quality throughout subsequent procedures. Cell counting in these kinds of samples becomes a challenging task due to common contaminants like red blood cells and non-cellular debris, introducing complexities such as time consumption, subjectivity, and a higher likelihood of errors.The EVE™ HT FL is a high-throughput automated fluorescence cell counter equipped with bright field and two fluorescence channels (AO/DAPI).AO (Acridine Orange) and DAPI (4′, 6-diamidino-2-phenylindole) are fluorescent dyes that bind to DNA, staining the nucleus of targeted cells. AO is a permeable dye that stains the nucleus of all cells, regardless of the cell’s condition. DAPI is an impermeable dye that only stains the nucleus of non-viable cells with damaged membranes. Basic principle of counting - Single reagent of EVE™ HT FL To accurately evaluate the viability of cell types like PBMCs or stem cells, it is advisable to use dual-fluorescence staining. This method effectively excludes non-nucleated cells (e.g., RBCs), platelets, and debris from the samples. In just 3 minutes, up to 48 samples can be counted and analyzed. How can EVE™ HT FL be applied in the manufacturing process of CAR-T?When working with cells like PBMCs or T-cells, accurate assessment of cell concentration and viability is essential for maintaining quality throughout subsequent procedures. Cell counting in these kinds of samples becomes a challenging task due to common contaminants like red blood cells and non-cell.1. Cell Viability Assessment:EVE™ HT FL, a high throughput fluorescence Cell Counter enable quick and accurate determination of the percentage of viable CAR-T cells, in other words, real-time monitoring of cell viability. Moreover, it allows for real-time monitoring of cell growth and proliferation. In CAR-T manufacturing, where a large number of T cells are often needed, this capability is crucial for for ensuring the overall quality of the final CAR-T product. By tracking these parameters, researchers can optimize culture conditions to ensure the production of a sufficient number of viable CAR-T cells.2. Enumeration of CAR-T Cells:EVE™ HT FL streamlines the enumeration process of CAR-T cells. By automating cell counting, it excel in enumerating total cell counts, including both viable and non-viable cells, further facilitating the determination of cell concentration. This allows for accurate seeding of cells during various stages of manufacturing process. This is crucial for maintaining the desired cell density and optimizing downstream processes.3. Quality Control of Starting Material:The success of CAR-T therapy is highly dependent on the quality of the starting T cell population. EVE™ HT FL facilitate a comprehensive evaluation of cellular morphology, size, and overall quality. Ensuring that the starting T cells meet predetermined criteria is essential for consistent and effective CAR-T production.4. Optimization of Culture Conditions:Continuous monitoring of cell cultures provides insights into the effects of different culture conditions on CAR-T cell growth and health. Researchers can use this information to optimize media composition, incubation parameters, and other variables, leading to enhanced CAR-T cell production efficiency.5. Determination of Optimal Harvesting Time:EVE™ HT FL aid in determining the optimal harvesting time for CAR-T cells. By monitoring cell viability during the culture period, researchers can identify the point at which the cells exhibit maximum viability and are most suitable for harvesting, optimizing the overall manufacturing timeline.6. Detection of Contaminants or Abnormalities:AO/DAPI staining can highlight contaminants or abnormalities within the cell population. Deviations in cell morphology or the presence of unwanted cell types can be quickly identified, allowing for corrective actions to be taken to maintain the purity of the CAR-T cell product.ConclusionIn conclusion, EVE™ HT FL can be applied in CAR-T manufacturing, offering capabilities for assessing cell viability, enumerating cell counts, monitoring cell growth, and ensuring the overall quality of the CAR-T cell product. Its high-throughput nature contributes to the efficiency and success of the manufacturing process, ultimately impacting the therapeutic efficacy of CAR-T cell therapies.

-
Revolutionizing Cell Counting: NanoEntek's Evolution from Hemocytometer to Cutting-edge Innovations
Pioneering a New Era in Cell Counting Technology Introduction: Cell counting has been a cornerstone in scientific and medical research, providing critical insights into biological processes and cell viability. The journey began with Louis-Charles Malassez's invention of the hemocytometer, a groundbreaking device that laid the foundation for cell counting. However, as technology advanced, NanoEntek emerged as a key player, revolutionizing the process and simplifying cell counting through a series of innovative products. The Origin of Cell Counting and Development: Hemocytometer Louis-Charles Malassez's hemocytometer marked the inception of cell counting, offering a precise method for counting blood cells. The Neubauer counting chamber became a standard tool in laboratories. This traditional method, while effective, presented inconveniences that inspired NanoEntek to pioneer advancements.Hemocytometer Counting Grid NanoEntek’s Continuous innovation towards Cell counting1) NanoEntek’s innovation in Simplifying Cell Counting: C-chip In response to the inconvenience associated with traditional glass slide hemocytometer, NanoEntek invented ‘C-chip’ - a disposable plastic hemocytometer. The C-chip eliminated the need for washing and cover slips, streamlining the cell counting process while maintaining accuracy. This innovation was a pivotal step in simplifying and modernizing cell counting techniques. 2) NanoEntek’s Innovation Continues: EVE™, EVE™ Plus NanoEntek introduced faster and more accurate automated cell counters, namely 'EVE™' and 'EVE™ Plus.' These devices not only expedited the counting process but also incorporated automated image analysis, enhancing the assessment of cell viability. The presentation of data in graphical formats further improved user convenience, allowing for more comprehensive and insightful analysis. NanoEntek’s Product Line-up for Cell Counting1) Automated Cell counter NanoEntek's product lineup for cell counting includes the C-chip (2ch/4ch), a disposable hemocytometer known for its high accuracy and time-saving features. EVE™ Plus stands out as one of the fastest cell counters, capable of counting cells within 1 second. For precise cell counting, ADAM™ MC2 utilizes PI staining technology. Notably, ADAM™ CellT meets the regulatory standards of 21CFR Part 11, making it suitable for use in cGMP production environments. NanoEntek's automated cell counters 1) Automated Cell counter NanoEntek's product lineup for cell counting includes the C-chip (2ch/4ch), a disposable hemocytometer known for its high accuracy and time-saving features. EVE™ Plus stands out as one of the fastest cell counters, capable of counting cells within 1 second. For precise cell counting, ADAM™ MC2 utilizes PI staining technology. Notably, ADAM™ CellT meets the regulatory standards of 21CFR Part 11, making it suitable for use in cGMP production environments. 2) High-throughput automated cell counter(1) EVE™ HTEVE™ HT is an automated cell counter that uses trypan blue for high-throughput measurements.In less than 3 minutes, it can accurately measure up to 48 samples, making it an efficient solution for busy laboratories or industries. The efficiency is maximized through the use of a disposable 48 channel plate. For those in regulated environments, EVE™ HT offers an optional 21CFR Part11 compliance, ensuring data integrity and security.EVE™ HT(2) EVE™ HT FLEVE™ HT FL is a high-throughput dual fluorescence cell counter.Equipped with a bright field and two fluorescence channels, namely AO and DAPI, EVE™ HT FL exceeds in speed and accuracy. In fact, its accuracy is evidenced with the R squared value of 0.99 it shows when compared to a flow cytometer. EVE™ HT FL can accurately measure up to 48 samples in less than 3 minutes, making it an efficient solution for busy laboratories or industries. The efficiency is maximized through the use of a disposable 48-channel plate.Remarkably, EVE™ HT FL only requires 20µL of sample per each measurement, which allows users to substantially reduce sample consumption by up to 25 times less than the competing instruments. This not only promotes efficient usage of the samples, but also let users to preserve their precious samples for additional evaluations or experiments.EVE™ HT FL offers an outstanding high-throughput cell counting method suitable for research and development in bio-processing and cell therapies. Its combination of speed, accuracy, and sample efficiency makes it a valuable tool in various scientific applications.EVE™ HT FLNanoEntek’s InnovationFor the past 20 years, NanoEntek has consistently led in innovation, introducing creative and groundbreaking products. Our dedication to sharing inventive ideas for the continuous development of our world is unwavering. Through a relentless pursuit of creativity, we aim to make meaningful strides in enhancing our global community.


 KOR
KOR ENG
ENG